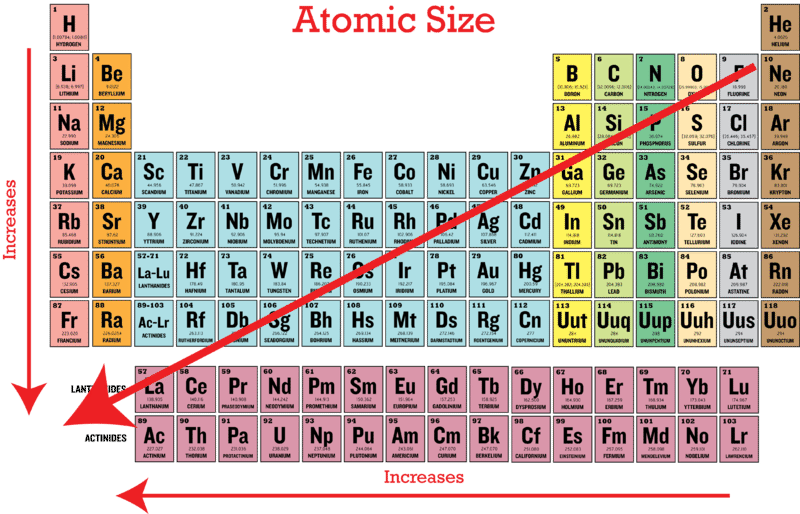
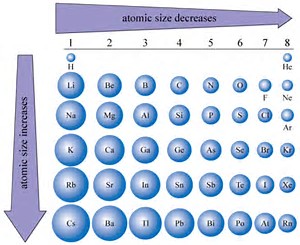
Why does Hydrogen, with just one electron, have a bigger atomic radius than other elements such as Neon and Oxygen? - Quora

Warm Up Where are the biggest atoms on the periodic table? 2.Arrange the following elements in increasing electronegativity: Cl, As, Sn 3.Which. - ppt download

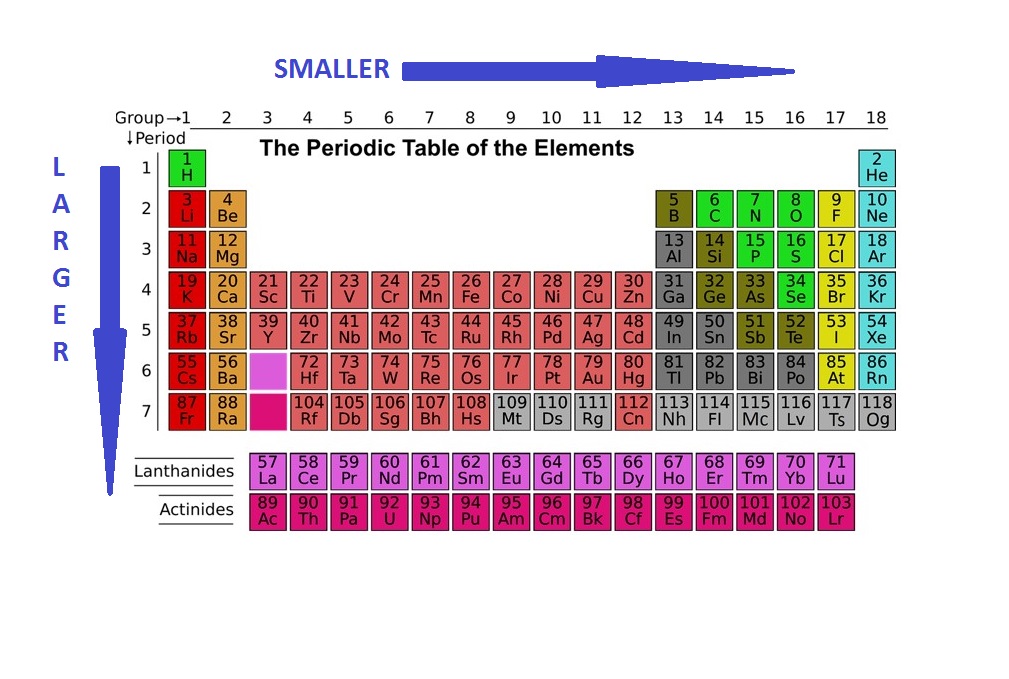
/PeriodicTable_AtomSizes-56a131193df78cf772684720.png)