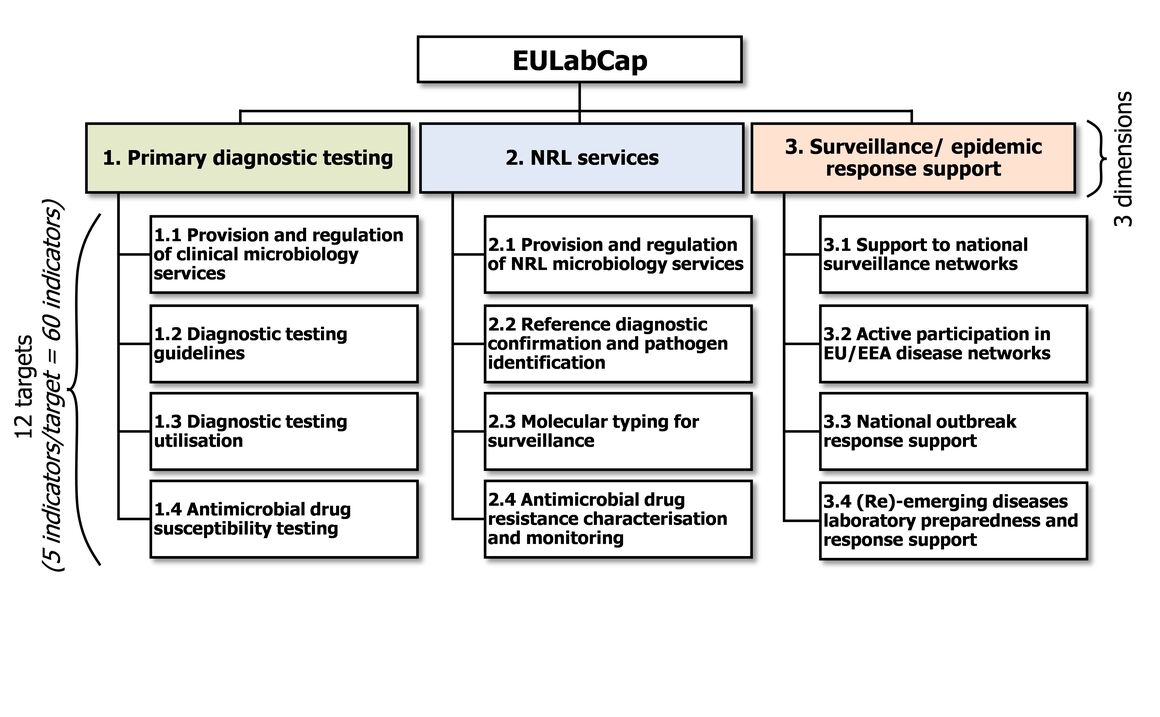
EU Laboratory Capability Monitoring System (EULabCap): Report on 2016 survey of EU/EEA country capabilities and capacities

Lifecycle Management for Near-Sterile Facility Contamination Control Programs | American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology
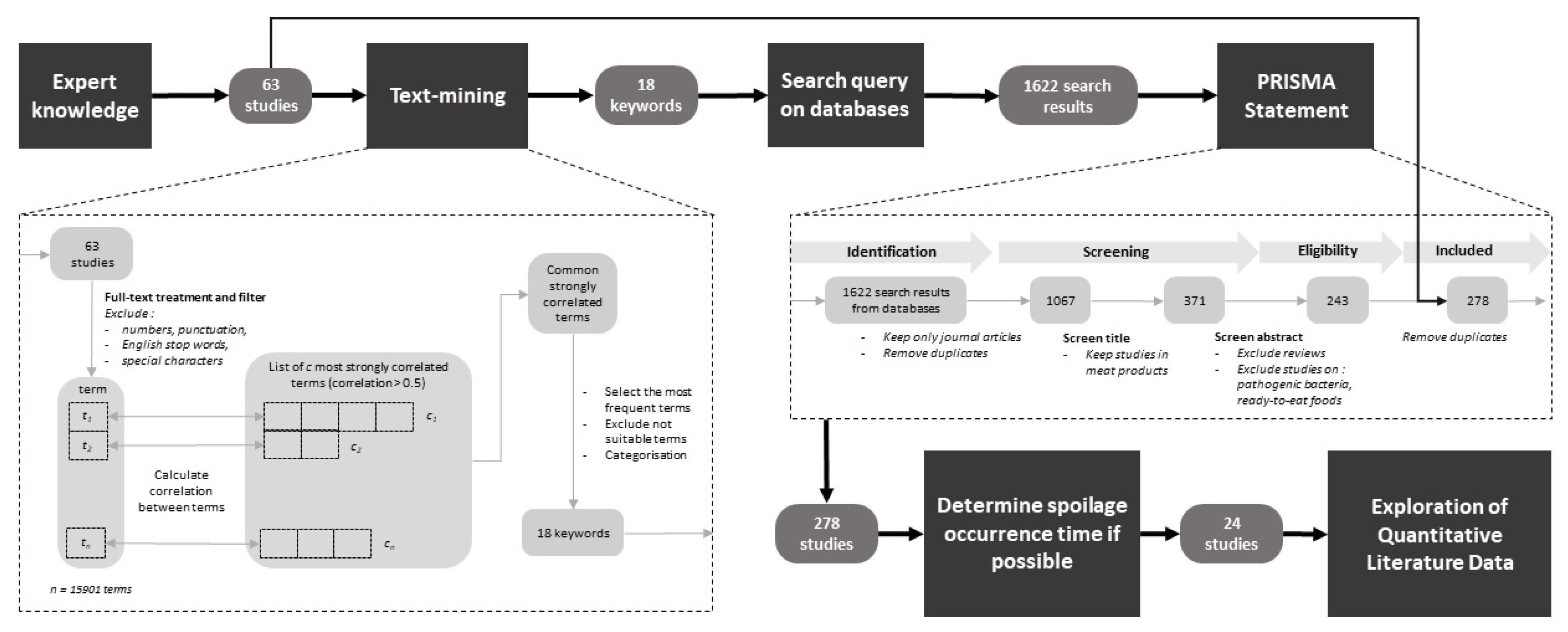
Microorganisms | Free Full-Text | Spoilage of Chilled Fresh Meat Products during Storage: A Quantitative Analysis of Literature Data | HTML

PDF) A Pilot Study for Monitoring Changes in the Microbiological Component of Metalworking Fluids as a Function of Time and Use in the System

Microbial aspects - Guidelines for Drinking-Water Quality: Fourth Edition Incorporating the First Addendum - NCBI Bookshelf

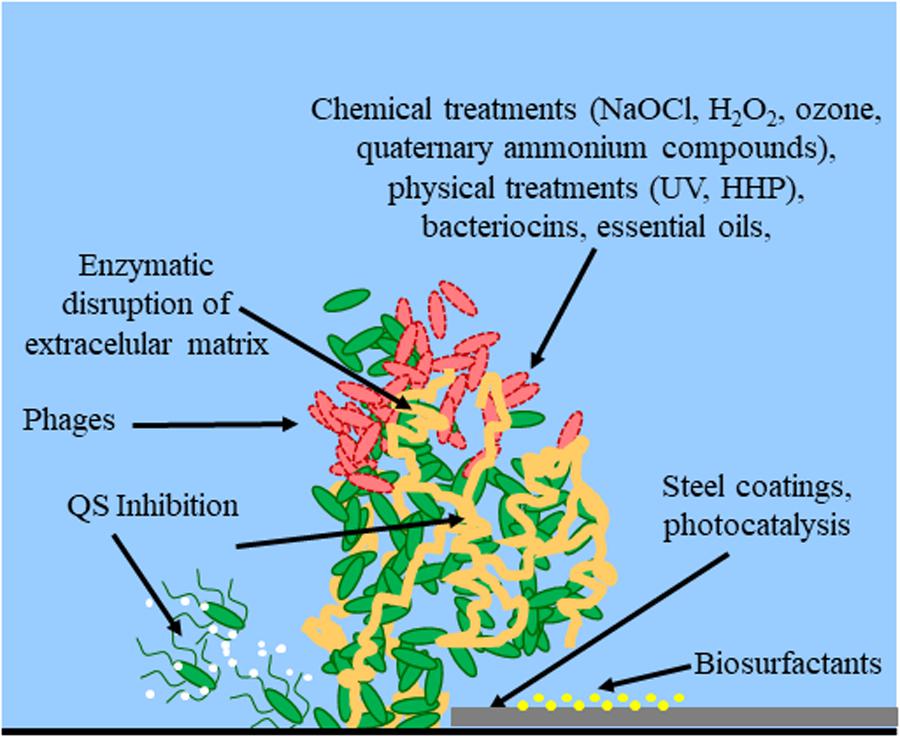
How to carry out microbiological sampling of healthcare environment surfaces? A review of current evidence - ScienceDirect

Case Studies of Microbial Contamination in Biologic Product Manufacturing | American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology

Role of Environmental Monitoring and Microbiological Testing During Manufacture of Sterile Drugs and Biologics* | American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology