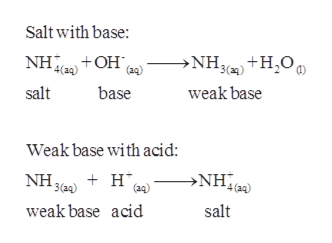Calculate the amount of NH3 and NH4Cl required to prepare a buffer solution of pH = 9 when total concentration of buffering - Sarthaks eConnect | Largest Online Education Community

Determination of 1 × 10 −5 M Co(III) with standard additions ((b), (c),... | Download Scientific Diagram

Calculate the pH of 0.5 L of a 0.2 M NH4Cl - 0.2 M NH3 buffer before and after addition of (a) 0.05 mole of NaOH and (b) 0.05 mole of HCI.
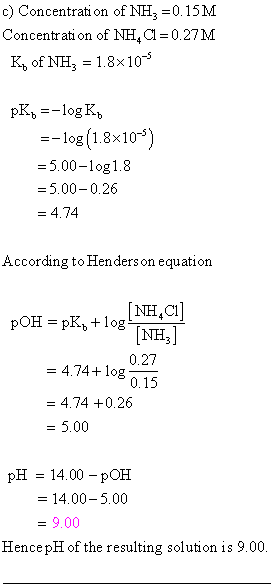
OneClass: A)Calculate the pH of the 0.20 M NH3/0.24 M NH4Cl buffer. B)What is the pH of the buffer af...

OneClass: Calculate the pH of a .20M NH3/.20M NH4Cl buffer after the addition of 20.0mL of 0.10M HCl ...












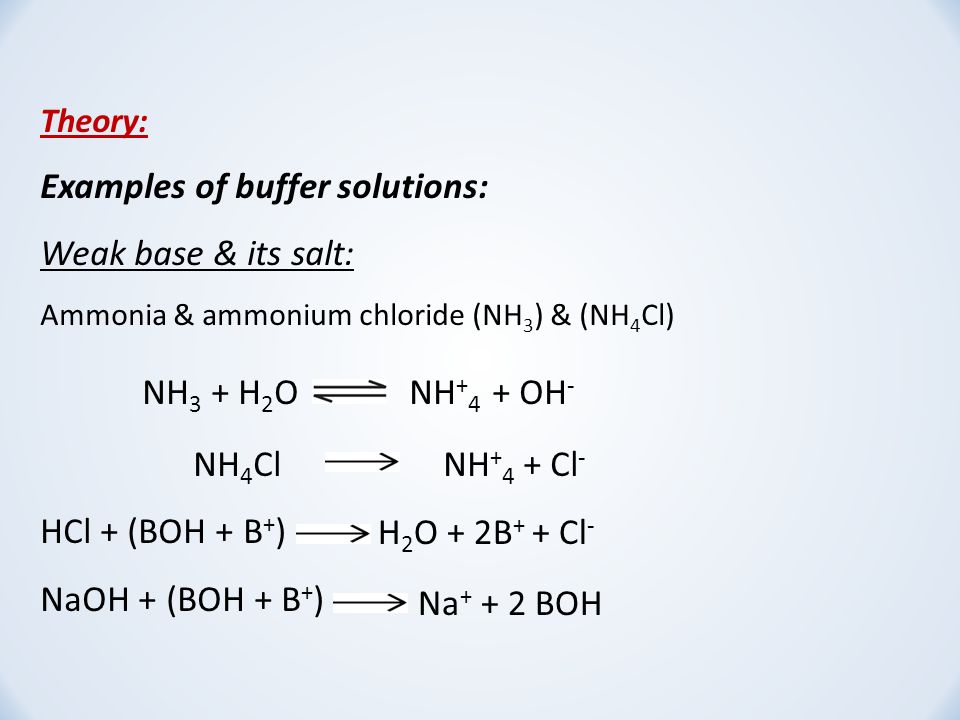
![Solved: BUFFERS 10. An Ammonium[NH3]/ammonium Chloride (NH... | Chegg.com Solved: BUFFERS 10. An Ammonium[NH3]/ammonium Chloride (NH... | Chegg.com](https://media.cheggcdn.com/media/98f/s907x655/98fc93a3-c6f6-4a89-8acb-45d7d9f5eb4d/image.png)