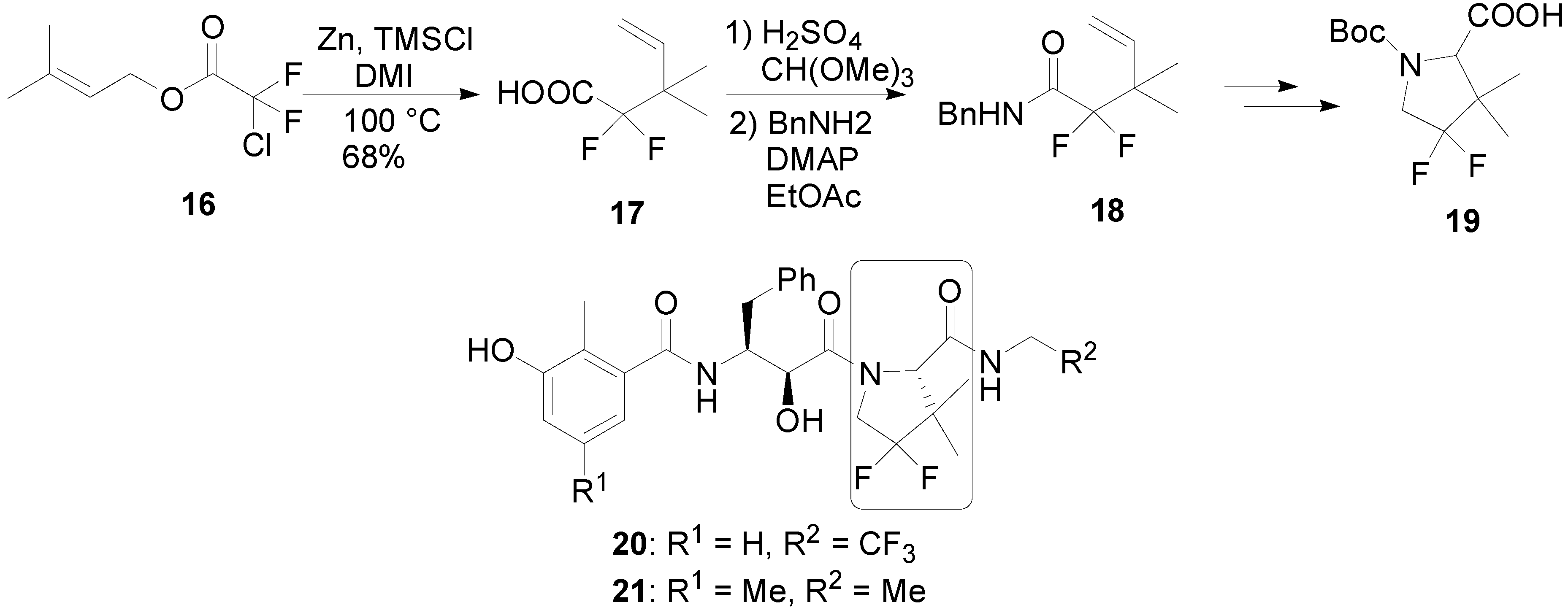![PDF) A new palladium(II)-catalyzed [3,3] aza-Claisen rearrangement of 3-allyloxy-5-aryl-1,2,4-oxadiazoles | Antonio Piccionello - Academia.edu PDF) A new palladium(II)-catalyzed [3,3] aza-Claisen rearrangement of 3-allyloxy-5-aryl-1,2,4-oxadiazoles | Antonio Piccionello - Academia.edu](https://0.academia-photos.com/attachment_thumbnails/44982140/mini_magick20190213-14539-zxuyqy.png?1550072610)
PDF) A new palladium(II)-catalyzed [3,3] aza-Claisen rearrangement of 3-allyloxy-5-aryl-1,2,4-oxadiazoles | Antonio Piccionello - Academia.edu

Asymmetric Synthesis of Allenyl Oxindoles and Spirooxindoles by a Catalytic Enantioselective Saucy–Marbet Claisen Rearrangement - Cao - 2012 - Angewandte Chemie International Edition - Wiley Online Library
![Palladium-catalyzed sequential reaction viaSonogashira coupling, isomerization, Claisen rearrangement and [4 + 2] cycloaddition sequence for the rapid synthesis of tricyclo[3.2.1.02,7]oct-3-ene derivatives - RSC Advances (RSC Publishing) Palladium-catalyzed sequential reaction viaSonogashira coupling, isomerization, Claisen rearrangement and [4 + 2] cycloaddition sequence for the rapid synthesis of tricyclo[3.2.1.02,7]oct-3-ene derivatives - RSC Advances (RSC Publishing)](https://pubs.rsc.org/en/Content/Image/GA/C1RA00452B)
Palladium-catalyzed sequential reaction viaSonogashira coupling, isomerization, Claisen rearrangement and [4 + 2] cycloaddition sequence for the rapid synthesis of tricyclo[3.2.1.02,7]oct-3-ene derivatives - RSC Advances (RSC Publishing)

Palladium(II)‐Catalyzed CH Activation/CC Cross‐Coupling Reactions: Versatility and Practicality - Chen - 2009 - Angewandte Chemie International Edition - Wiley Online Library
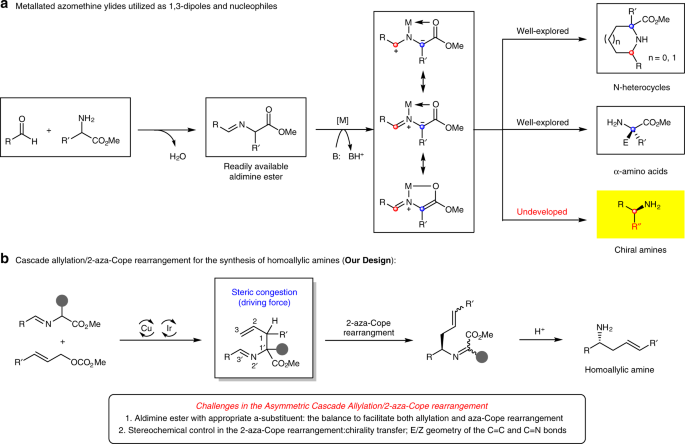
Synergistic catalysis for cascade allylation and 2-aza-cope rearrangement of azomethine ylides | Nature Communications

Synthesis of (±)‐epi‐Jungianol by the Gold(I)‐Catalyzed Propargyl Claisen Rearrangement/Hydroarylation Cascade Reaction of Propargyl Vinyl Ethers - Rinaldi - 2021 - European Journal of Organic Chemistry - Wiley Online Library
![Palladium-catalyzed sequential reaction via Sonogashira coupling, isomerization, Claisen rearrangement and [4 + 2] cycloaddition sequence for the rapi ... - RSC Advances (RSC Publishing) DOI:10.1039/C1RA00452B Palladium-catalyzed sequential reaction via Sonogashira coupling, isomerization, Claisen rearrangement and [4 + 2] cycloaddition sequence for the rapi ... - RSC Advances (RSC Publishing) DOI:10.1039/C1RA00452B](https://pubs.rsc.org/image/article/2012/RA/c1ra00452b/c1ra00452b-s1.gif)
Palladium-catalyzed sequential reaction via Sonogashira coupling, isomerization, Claisen rearrangement and [4 + 2] cycloaddition sequence for the rapi ... - RSC Advances (RSC Publishing) DOI:10.1039/C1RA00452B

Toward a symphony of reactivity: cascades involving catalysis and sigmatropic rearrangements. - Abstract - Europe PMC

Figure A93 from Part A: Palladium(II)-catalyzed enantioselective Saucy-Marbet Claisen rearrangement of propargyloxy indoles to quaternary oxindoles and spirocyclic lactones. Part B: The regioselective oxidative coupling of phenols | Semantic Scholar

Catalysis of the Claisen Rearrangement of Aliphatic Allyl Vinyl Ethers - Hiersemann - 2002 - European Journal of Organic Chemistry - Wiley Online Library
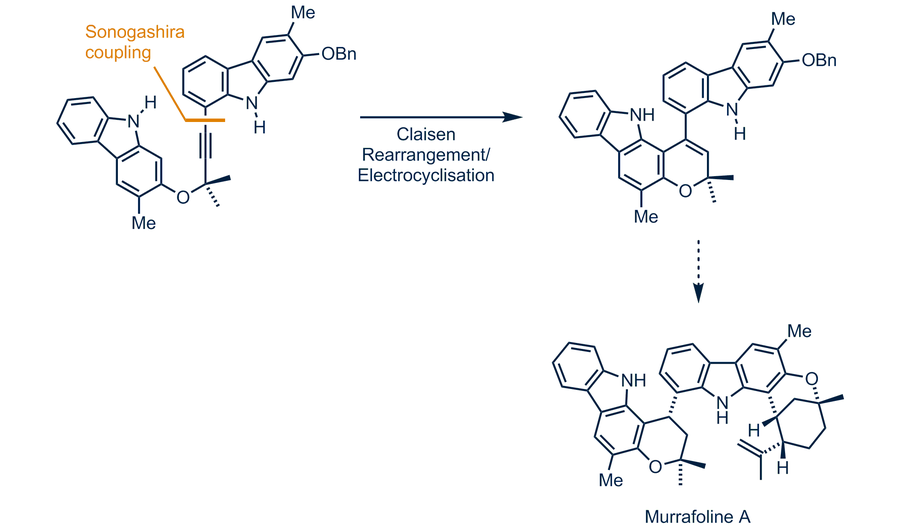
Palladium(II)-catalyzed synthesis of carbazole alkaloids — Chair of Organic Chemistry II — TU Dresden

Asymmetric aza-Claisen rearrangement of allyl imidates catalyzed by homochiral cationic palladium(II) complexes - ScienceDirect





![Base-Mediated Claisen Rearrangement of CF3-Containing Bisallyl Ethers[v1] | Preprints Base-Mediated Claisen Rearrangement of CF3-Containing Bisallyl Ethers[v1] | Preprints](https://www.preprints.org/img/dyn_abstract_figures/2021/06/f01e66a84543cbcea2fb69e0d6305f24/preprints-44888-graphical.v1.png)